Hera Biolabs’ piggyBac transposase is proven to rapidly create stable in vivo reporter cell lines for imaging tumor growth and metastasis. Hera has the piggyBac cell line engineering capabilities, unique oncology rodent models and now has the Spectral Instruments Ami HT optical imaging system as well for advanced in vivo imaging.
Let’s review a recent study that used the piggyBac transposon system to engineer a human gastric adenocarcinoma cell line BGC823-iRFP720-GFP (abbreviated as BGC823-iRFP) that stably expressed iRFP720 and green fluorescent protein (GFP). Tracing techniques based on iRFP720 may optimize the noninvasive monitoring of tumors in vivo. Overall, IVIS Lumina III imaging indicated that the iRFP720 signals of the BGC823-iRFP cells could be used to evaluate the antitumor efficacy of oncolytic viruses and chemotherapy drugs.
Background
One of the most common forms of digestive tract malignancies is gastric cancer (GC), and many investigations have been focused on finding novel therapeutics and utilizing the science behind the invasion and metastasis mechanism of GC. In many cases, luciferase, a bioluminescent enzyme, is used to adapt GC cells for in vivo optical imaging.
There have been several series of iRFPs (NIR fluorescent proteins) that have been developed for in vivo imaging (Shcherbakova and Verkhusha, 2013; Chernov et al., 2017; Oliinyk et al., 2019). One variation, iRFP720 has recently been validated to be easily detected in vivo because of its minimal absorption in mammalian tissues. So far it has been used to mark ovarian cancer, lung cancer, and breast cancer (Rice et al., 2015; Geyer et al., 2017; Comenge et al., 2018; Wilson et al., 2019).
Materials And Methods
Regarding the plasmid DNA, the iRFP gene was synthesized by GenScript Biotech and inserted into the plasmid piggyBac Dual Promoter (pPBDP). The helper vector, Super piggyBac Transposase expression plasmid (pSPBT), was obtained from System Biosciences. If you would like to see more details regarding the materials and methods used in this study, please refer here.
Development Of BGC823-iRFP Cell Line
For generation and characterization of the cell line, four iRFP720-GFP-positive subclones were initially isolated, and then the subclone with the strongest fluorescence intensity was selected for the experiments and was subsequently named BGC823-iRFP cell line. BGC823-iRFP had similar characteristics to the BGC823 parent line both in vitro and in vivo. 6 week old nude mice were subcutaneously injected with either the BGC823 line or the edited BGC823-iRFP line (see figure B). On day 18, the mouse intraperitoneally injected with BGC823-iRPF showed obvious signals (see figure E).

In Vivo Evaluation Of BGC823-iRFP
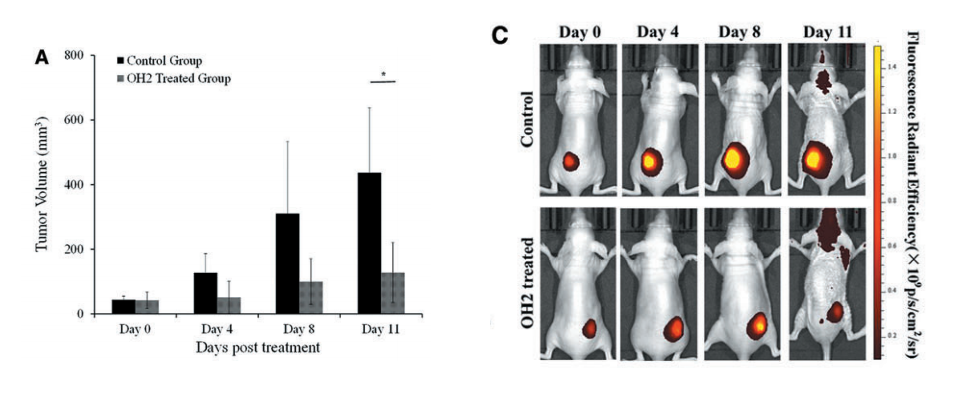
As you can see in Figure C (below), the initial iRFP720 fluorescence intensity observation exhibited a clear ‘‘hotspot’’ at the BGC823-iRFP injection site. The mice were treated with OH2 virus (a genetically engineered virus designed to enhance tumor-specific immune responses). On day 4, the fluorescence signals significantly enhanced and enlarged in the control mice group, strongly suggesting that the tumor grew with the proliferation of BGC823-iRFP cells. The fluorescence signals in the OH2-treated group of mice were consistently lower than those in the control group (Figure A). The fluorescence images were acquired using IVIS Lumina III equipped with 690 nm excitation and 713 nm emission filters.
Hera Biolabs Spectral Instruments Optical Imaging System
Hera Biolabs has been working on developing a platform using PiggyBac to rapidly create in vivo reporter cell lines for imaging tumor growth and metastasis. The piggyBac platform is very efficient for creating reporter cell lines. Hera has these capabilities and now has the Spectral Instruments Ami HT system for advanced in vivo imaging. We are excited to have the Spectral Instruments system in our laboratory as well as the new capabilities it brings.
The highly robust SRG™ OncoRat® may present several advantages for Hera’s partners, especially for translational imaging studies with orthotopic tumors. Mouse orthotopic tumor studies have demonstrated clinically accurate tumor microenvironment and vasculature, but implantation can be difficult due to the very small size. A recent protocol published in JOVE “A Bioluminescent and Fluorescent Orthotopic Syngeneic Murine Model of Androgen-dependent and Castration-resistant Prostate Cancer” highlights orthotopic injection and results in mice.
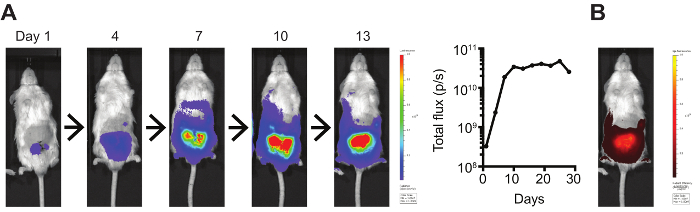
Orthotopic Myc-CaP tumors stably expressing luciferase were imaged using an IVIS Spectrum Imaging System. Bioluminescence was quantified by total flux (photons/s)
“At Hera we are very excited to apply non-invasive imaging technology to our in vivo models. We will be broadening our client services to include subcutaneous, orthotopic, and metastatic analysis in both immunodeficient mouse and rat.” -Study Director, Hera BioLabs
If you would like to learn more about the imaging technology and Hera’s CRO services, please contact us here today.
