Gene editing technology continues to shape drug discovery and development research. Scientists utilize these tools to not only edit DNA for purposes of cell and gene therapies, but also to study disease states and their molecular targets and pathways.
We help researchers create cellular models to mimic and study diseases, and screen for the next therapeutic breakthrough. The combination of Cas-CLOVER™ for targeted editing and piggyBac® transposase for stable expression of any sized DNA enables unlimited capacity for model creation.
Partner with us for experience in over 75 cell lines or order reagents, the choice is yours.
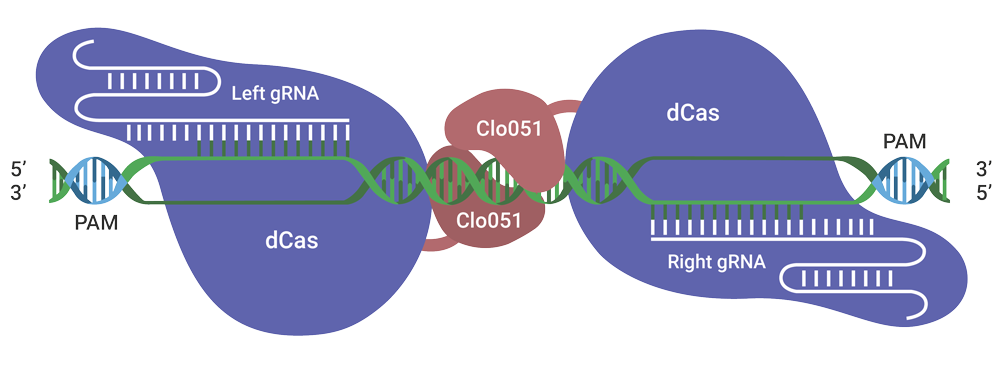
Targeted And Effective Gene Editing With Cas-CLOVER
One of our key gene editing tools is Cas-CLOVER, a cleaner CRISPR alternative, which serves as “molecular scissors” to introduce targeted double-strand breaks in genomic DNA for knockouts, knock-ins and base-pair edits.
Such tools have a wide range of drug discovery and development applications including cell-line engineering, and animal model creation.
Cas-CLOVER More Precise Than CRISPR
Cas-CLOVER is unique compared to other targeted gene editing tools as it combines the efficiency of CRISPR/Cas9 with the specificity of TALENs:
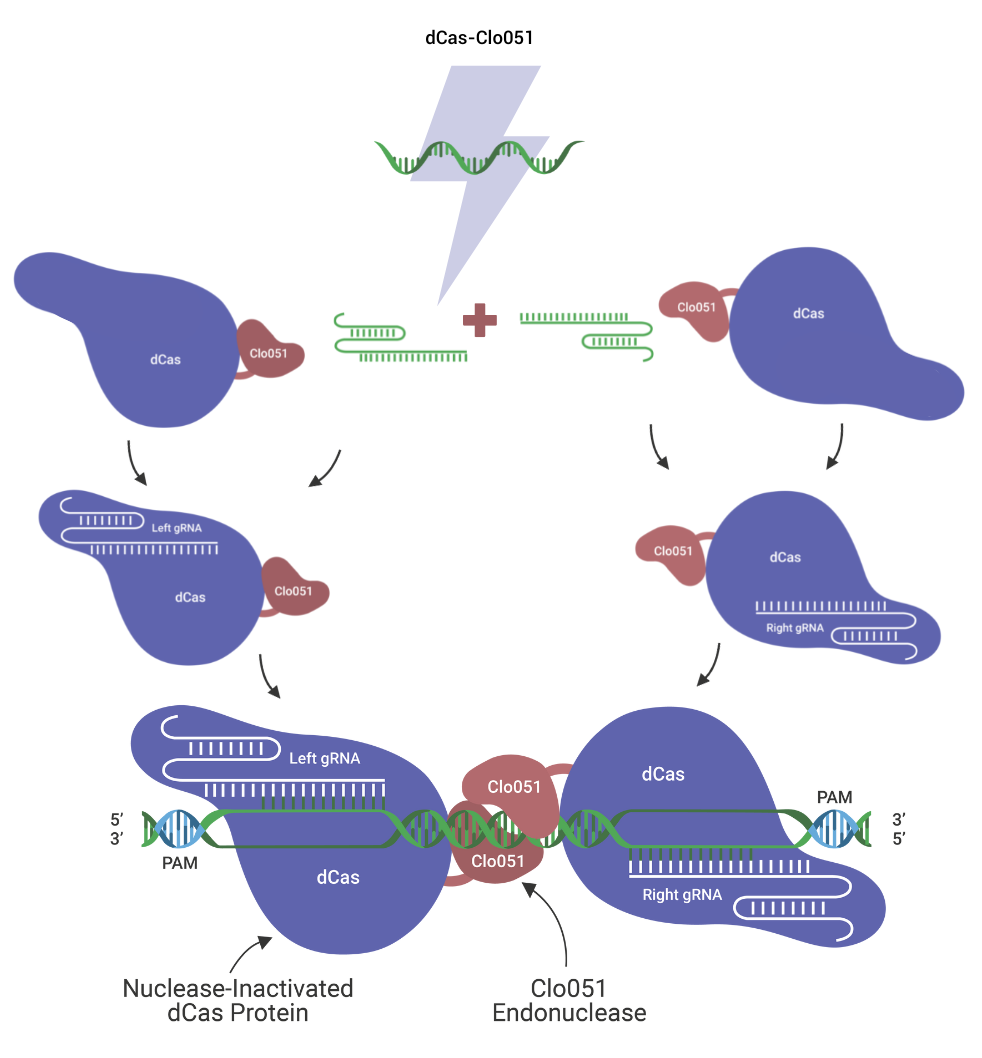
High efficiency: Cas-CLOVER is recruited to the target site by guide RNAs (gRNAs) in a similar way as Cas9. However, the Cas9 is completely deactivated “dCas9” and only serves as a linker to our high efficiency proprietary dimeric nuclease Clo51.
High specificity: Cas-CLOVER utilizes two gRNAs which recruit two dCas9-Clo51 nucleases to initiate targeted cleavage. The dCas9-Clo51 nuclease is fully dimeric, meaning activity or cutting only occurs where a pair of nucleases bind correctly to the on-target genomic DNA. This technology has demonstrated best-in-class specificity by deep sequencing and Guide-Seq read-outs.
Easy to Use: The Cas-CLOVER technology can be integrated into any gene editing application or workflow with simple open-source design tools. With clear and independent issued intellectual property, Cas-CLOVER is simple, convenient, and accessible for R&D and commercial applications.
Large DNA Cargo Delivery & Footprint-Free Editing With piggyBac
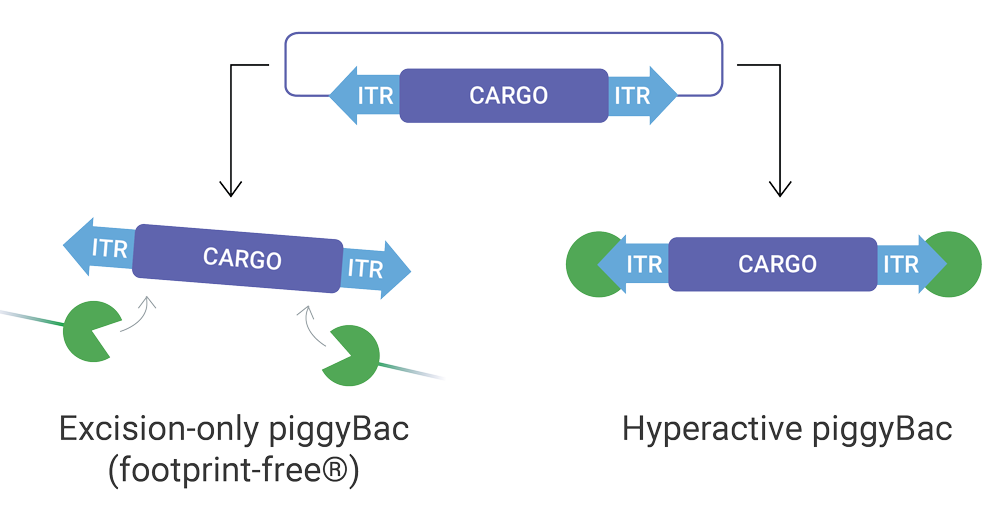
Need to create a stable cell line, transgenic animal or precise base-pair edit? piggyBac is a proven transposase + transposon DNA modification system which helps introduce small to very large genetic cargo stably into the genome, as well as remove genetic cargo in a scarless manner in transgenics and stable cell lines. piggyBac is non-viral, cost effective and very efficient.
Applications & Case Studies
Find related and up-to-date information and literature for our Cell Line Engineering service
including blogs, posters, presentations, webinars, and white papers.
Advanced Gene Editing Tools Made Accessible
The work products from our custom services typically come with a full research license. We are also offering clear commercial freedom to operate and simple accessible licenses for commercial applications in drug discovery.